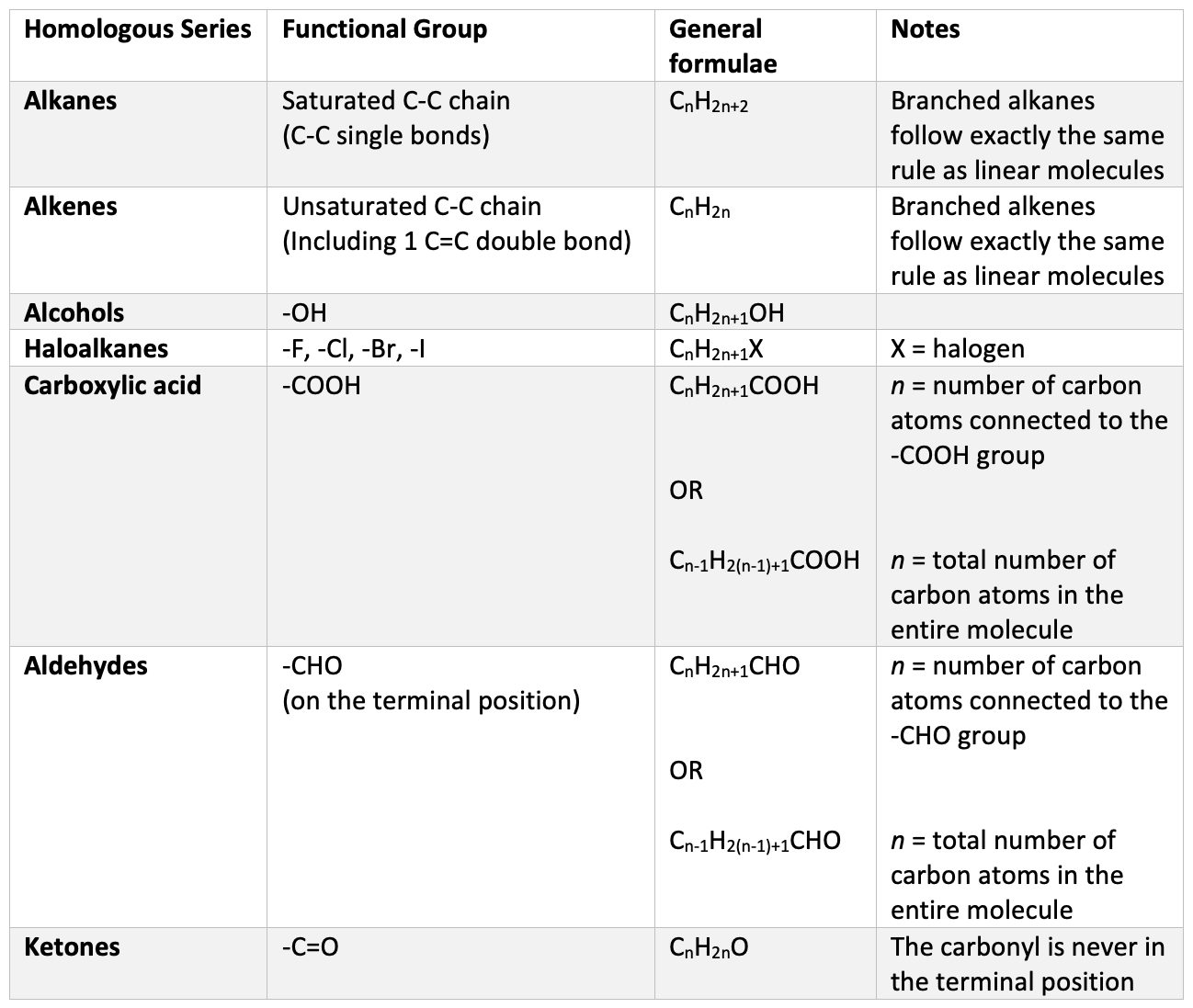
A common question to be asked in the exam, both in National 5 and Higher (and for GCSE and A-Level too) is to provide the general formula for a specific homologous series.
This type of question can come in two forms: specifying the homologous series in question and asking, “what is the general formula for the alcohols?”; or showing you the structure of a molecule and asking you to “write the general formula for the homologous series to which this molecule belongs”, or something similar.
The first type of question is just testing your memory of all the different homologous series that you have encountered – simply recalling the general formula (CnH2n+1OH if you were wondering).
In the second type above, you are challenged to recognise what homologous series the molecule belongs to first. A variation of this might be “this hydrocarbon decolourises bromine water – what is its general formula?”. In this case, you need to deduce that decolourising bromine water is a key reaction (and identifying test) for an alkene – from this identification, you then state its general formula – CnH2n. Effectively in this type of question, your brain goes through a two-part process – recognition, followed by recall – applying it to the situation of the question.
These types of questions are actually very easy, however many pupils (and teachers dare I say!) forget the general formulae. This is quite understandable since there are a few, and they differ in somewhat counter intuitive ways.
If you are inclined to memorise them, this will provide you an excellent advantage for this type of question, as well as questions asking you to apply them. However, for those of us who struggle to remember them, understanding where the general formula actually comes from and being able to work them out for yourself is vital; and perhaps more beneficial for you in the long run than just simple memory work.
Working it out
Just what is a general formula? I think pupils often miss the point of them – thinking that they are just a simple curiosity associated with a homologous series; and something else to cram into revision before the exam. The general formula of a homologous series describes the relationship between the quantities of each element in the formula of all the molecules in that homologous series. No matter what the size is of the molecule, what structural isomer is being considered, all the molecules will fit the same general formula.
To illustrate this, let’s first consider the alkanes. Pictured below is butane. It has a chemical formula of C4H10. This means that it contains 4 carbon atoms and 10 hydrogen atoms. Before we look at how isomers of butane also possess this formula, lets first work out the general formula.

In a general formula, it is conventional to set the number of carbon atoms as the variable n. In butane’s case, n = 4. The general formula should allow us to calculate the number of hydrogen atoms also present in the molecule from the given number of carbon atoms. Let’s set this up:
CnHsomething
For butane :
C4Hsomething
Lets be a little more scientific than something – let’s say the number of hydrogen atoms is x :
C4Hx
We know from butane’s formula that x must be 10, so how do we make 10 from 4?
We could add 6 – but does that work?
CnHn+6
C(4)H(4+6) = C4H10
It obviously does for butane but let’s try it with another alkane – methane. We know that methane’s formula is CH4
C(1)H(1+6) = CH7 – This is in no way possible!
Simply adding 6 cannot work, so let’s try multiplying. We shall start with doubling:
CnH2n
C(1)H(2×1) = CH2 – still not right, but 2 is 2 away from 4, so let’s add 2:
C(1)H(2×1 +2) = CH4 – bingo! It works.
That gives us a potential general formula of CnH2n+2, but does it work with other molecules in this homologous series? Let’s return to butane (where n = 4) to check:
C(4)H(2×4 + 2) = C4H10 – It works!
And with octane? (which should be C8H18):
C(8)H(2×8 + 2) = C8H18
This confirms that our method is correct and that we have finally arrived at a working general formula.
Branching out
This works with single chain hydrocarbons, but does it also work for branched hydrocarbon chains?
Let’s take an isomer of butane – 2-methylpropane.
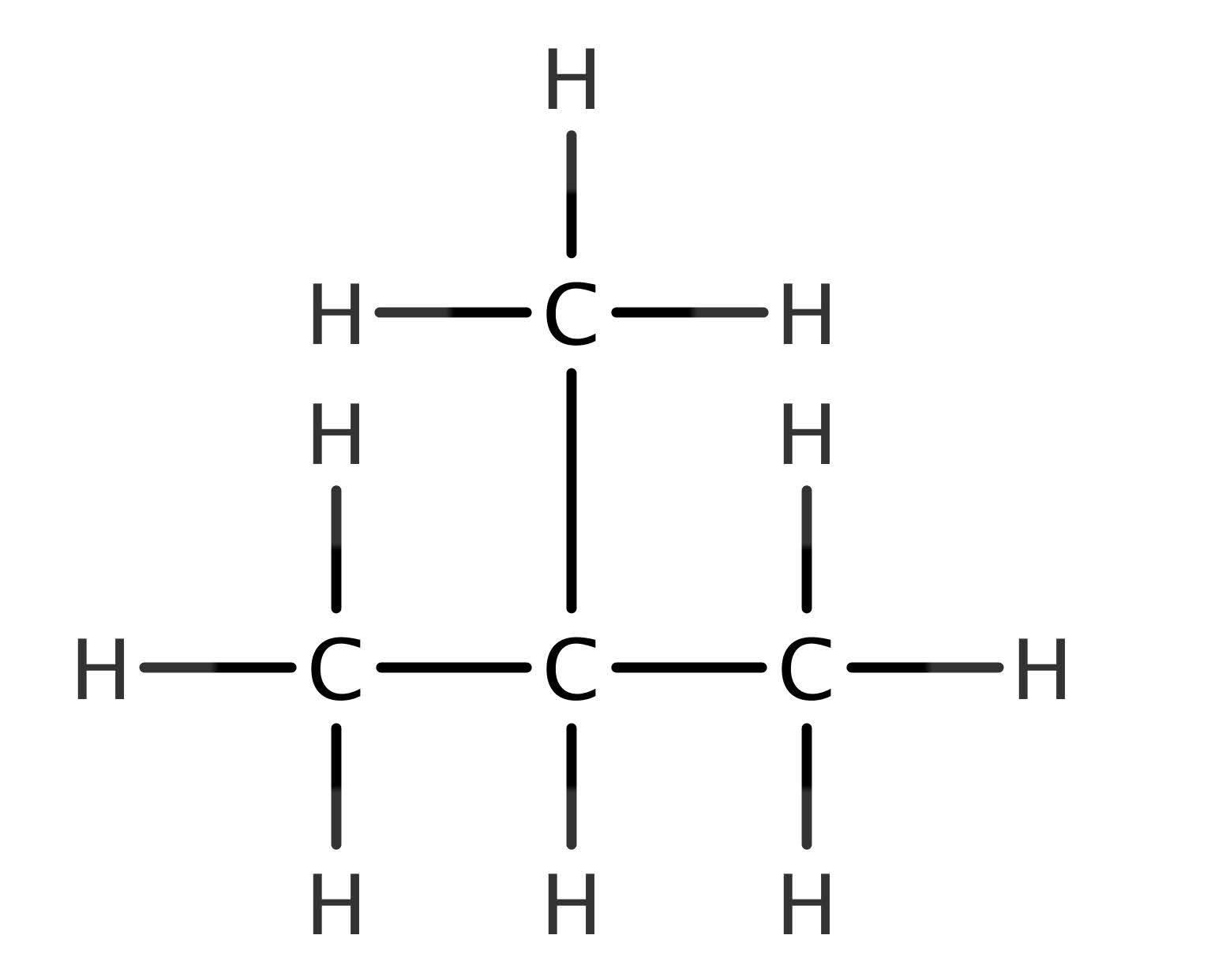
Does this molecule fit the same general formula of butane? An interesting question, one which we can answer in two different ways. Firstly, you could simply compare its formula with that of butane – if they match, then the answer must be yes. As we saw before, butane has a chemical formula of C4H10. To work out the formula of 2-methylpropane, simply count the number of carbon atoms and hydrogen atoms. It should be obvious that the formula of 2-methylpropane is also C4H10 – since this is the same as butane’s it must therefore have the same general formula as that of butane. An alternative way of approaching this question is to work out its general formula directly and comparing it to that of the alkanes. Since the formula is C4H10, this is of the form CnH2n+2, it therefore does indeed have the same general formula as butane.
Expanding to other functional groups
Something like the alkanes is easy to remember but being able to understand the process to work it out allows you to realise the logic in the concept; making it possible to apply this skill to other homologous series. Let’s look at the alcohols. The general formula was mentioned above – CnH2n+1OH. This looks odd in comparison – you will have encountered alkanes, alkenes, and cycloalkanes, each with general formula involving just multiplying or a combination of multiplying and adding two, but this involves adding one. Where does that come from!? This is where most pupils struggle the most – going from the familiar to this slight deviation. Let’s compare two similar sized molecules to see the origins of this.
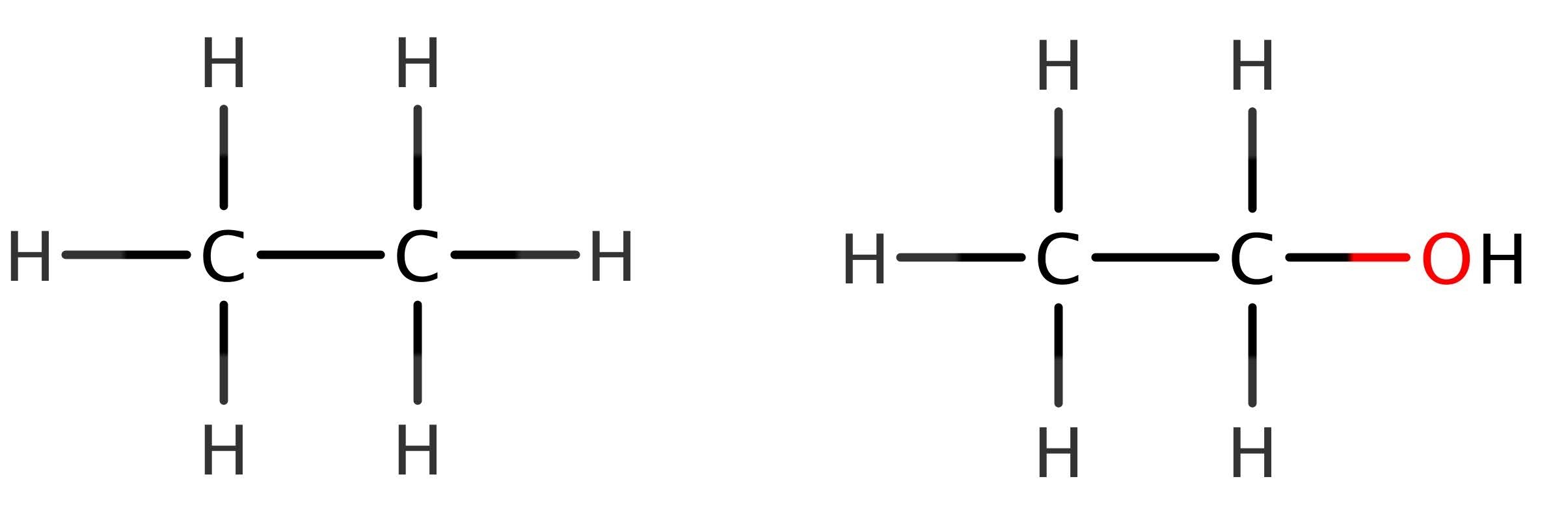
We do not have to look far to see where this comes from. Look at the above structural formulae. What is the difference? While the difference in the number of hydrogens in the general formulae may be counter intuitive, spotting the difference in the structure should be obvious! One of the hydrogens is replaced in the alcohol with a hydroxyl functional group. Therefore, there will be one less hydrogen in the general formula! (Note here that in total there are the same number of hydrogens present, but one is part of the hydroxyl group – compare this to a molecule with a halide instead of the hydroxyl group if you need to clarify this). This is where the 2n+1 comes from in the general formula – one hydrogen is swapped for the OH group, so we remove it appropriately from the equation.
Let’s experiment with this concept:
Propan-2-ol – does this have the same general formula as ethanol?

The formula is C3H7OH. How does 7 relate to 3? It’s 1 more than double it:
3 x 2 = 6, 6+1 = 7
Therefore, the general formula is CnH2n+1OH.
Does this fit with something more complex? A challenge now – what is the general formula of the homologous series to which 4‐ethyl‐3,4‐dimethylheptan‐2‐ol, belongs? Hopefully you know how to name branched chain hydrocarbons! If you can, then naming branched chain alcohols is exactly the same (I’ll write an article on naming and drawing structures later – pupils have endless fun with massively complex molecules once they get the hang of the naming process!). The longest chain is a heptane – so 7 carbons, 3 branches, an ethyl on the third to last carbon, then a methyl group on both of the next carbons, followed by the hydroxyl group on the second carbon atom from the other end.
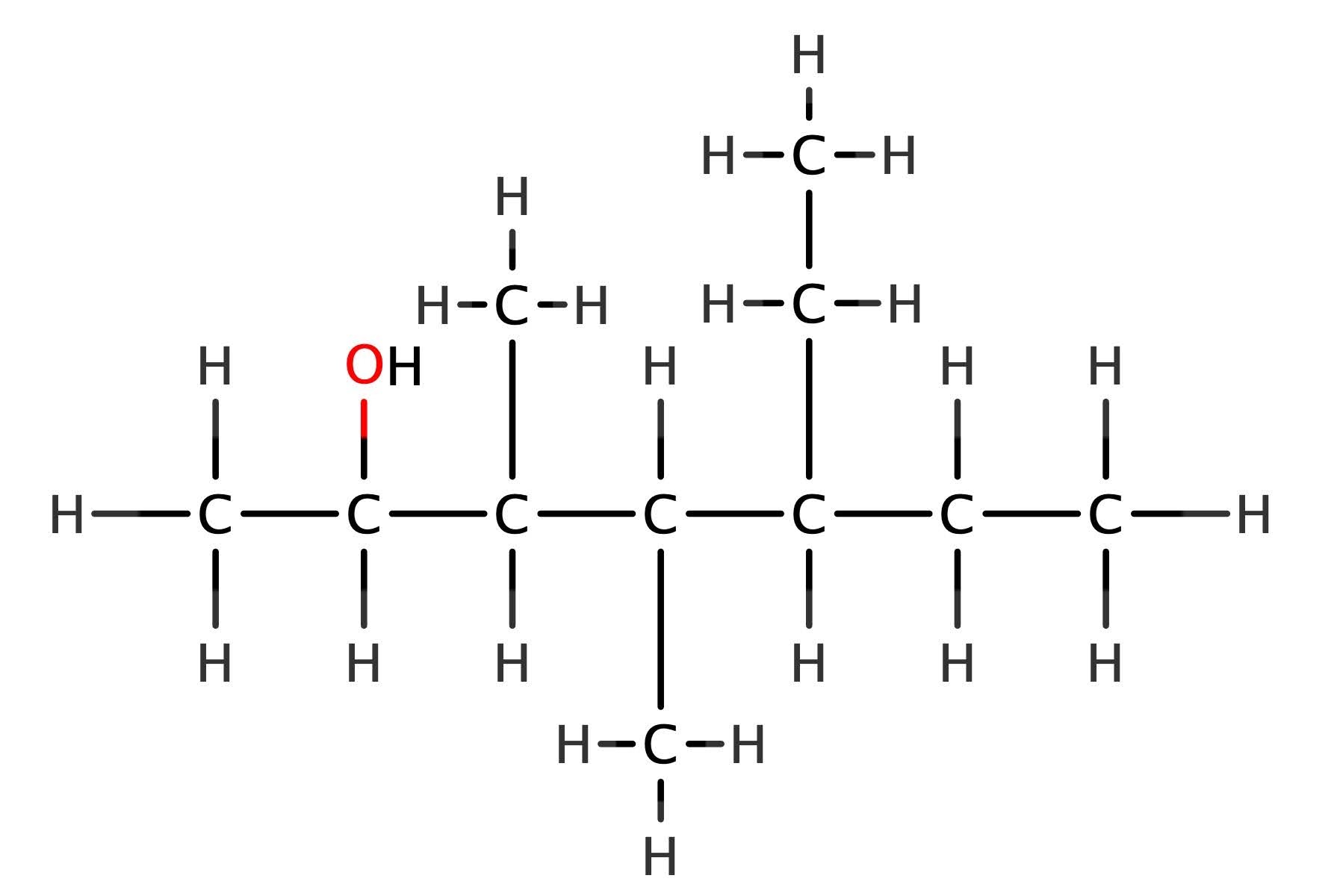
Does this have the same general formula as propan-2-ol? Let’s start by working out its formula – count each of the atoms present. The answer is C11H23OH (which incidentally is an isomer of undecanol). In order to answer our original question, lets take a closer look at those numbers. If n = 11 (the number of carbon atoms), how do we get to 23? Always start by doubling for general formula, as you may have noticed!
11 x 2 = 22
So how do we get to 23 from 22? Add 1!
11 x 2 + 1 = 23
So the general formula is indeed CnH2n+1OH. The answer is yes this does have the same general formula as propan-2-ol.
Involving the Halogens
Mentioned previously were the halides. Organohalides are not specifically part of the national 5 course, however they are mentioned; and you should be able to recognise them as addition products from alkenes (particularly the organobromides when bromine is added to an alkene).
Consider the structure of 1-chloropentane, which is an example of the chloroalkanes.
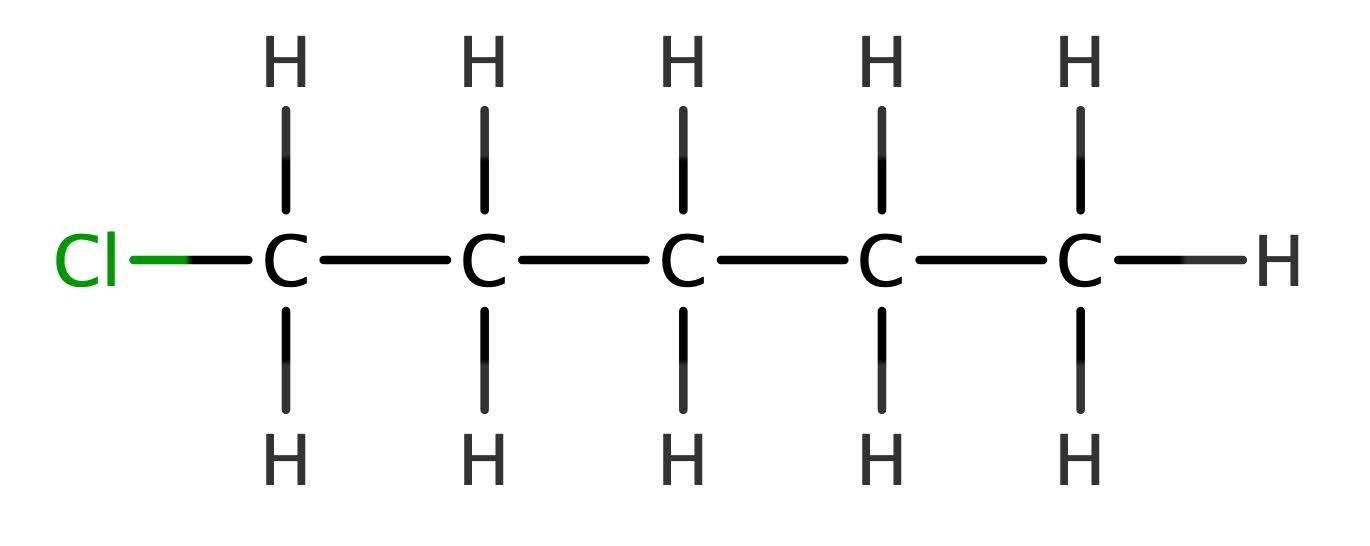
What does this remind you of? It bears a striking resemblance to an alcohol, with the hydroxyl group replaced by chlorine.
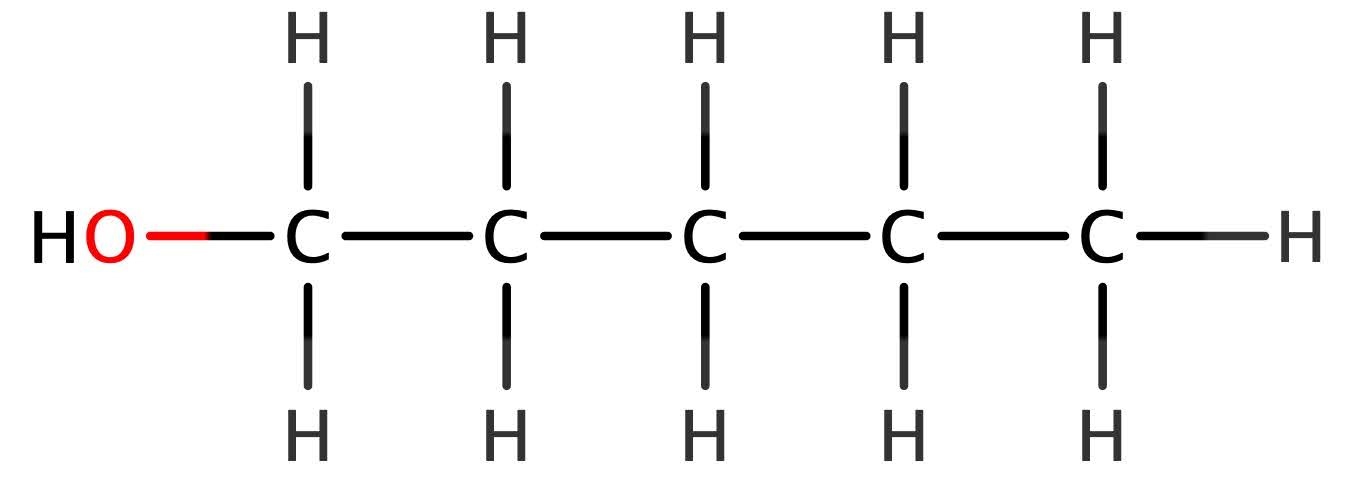
Therefore, obtaining its general formula must follow the same process. One hydrogen atom has been completely replaced by the chlorine, meaning that there is 1 less hydrogen than in the alkane equivalent molecule, pentane.

What is the general formula of the chloroalkanes? Start as always with working out the chemical formula of this example first. 5 carbon atoms, 11 hydrogens, and 1 chlorine, so the formula is C5H11Cl. What is the general formula? Well, let’s say that n = 5, how do we get to 11? Doubling it gives us 10 (5 x 2 = 10) which is 1 away from 11, so add 1. 5 x 2 + 1 = 11. This gives us a general formula of CnH2n+1Cl for the chloroalkanes. Exactly the same process could be applied to any of the organohalides – fluoroalkanes CnH2n+1F; bromoalkanes CnH2n+1Br; or iodoalkanes CnH2n+1I.
When working out the general formula, consider the similarities. In the 1-chloropentane example, the molecule is almost identical to that of pentane. Since one hydrogen is replaced by chlorine, the number of hydrogens decreases by 1 and is swapped by chlorine. If asked its formula, you could approach this by first giving the formula of pentane, C5H12, then modifying this to remove a hydrogen, C5H11, and finally adding on the chlorine atom, C5H11Cl.
Carbohydrate molecules
Special mention should go the carboxylic acids, aldehydes and ketones as they have some rather special chemistry, meaning that confusion often occurs with the actual number of carbon atoms in their structures.
Starting with the carboxylic acids, what is their functional group? This might seem odd at first, since the carbon atom on the end actually forms a part of this group. We have a hydroxyl group (-OH), but also a carbonyl (C=O). When both of these groups originate on a terminal position on the same carbon atom, that carbon is a part of that functional group, -COOH. My higher pupils affectionately referred to this as the “COOH group”, spoken with the sound a pigeon makes, as a way of remembering it (to each their own). The point being that the carbon is as much a part of this functional group as the oxygen and hydrogen atoms are. This means that the n in the general formula is actually only that of the chain minus that of the carbon attached to the functional group. It can be written two ways:
CnH2n+1COOH
Or
Cn-1H2(n-1)+1COOH
In the first form, n is equal to the number of carbons present in a chain attached to the carbon of the functional group. In the second form, n is the total number of carbons in the whole molecule – functional group and all. Let’s examine why this is. Take ethanoic acid. This has two carbon atoms.
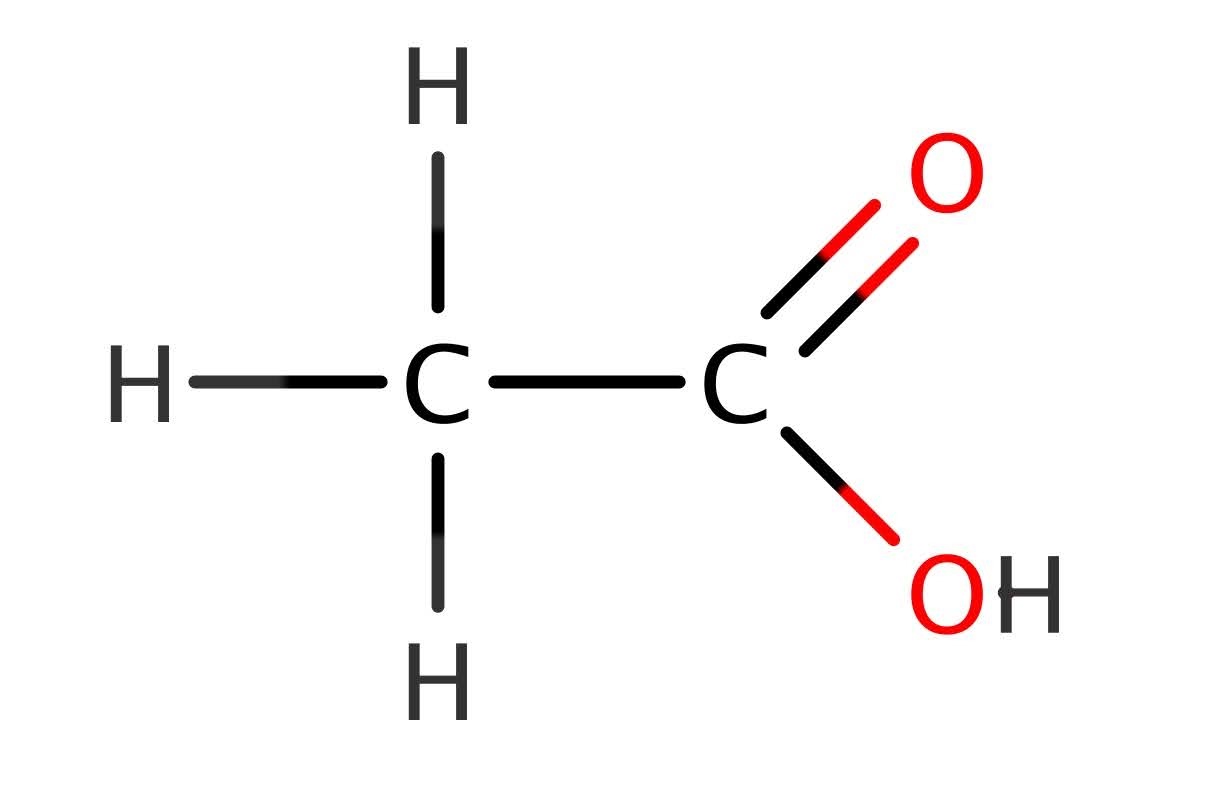
In the first formula, you have to be careful to take into account the COOH group. If you put the n = 2 into the formula, it will look like this :
C(2)H(2 x 2 + 1)COOH = C2H5COOH
This is wrong! It shows that there are 3 carbon atoms in ethanoic acid – we know from the structure (and the eth– prefix) that there should be 2!
Since there is 1 carbon atom already in the COOH group, this means that there is only 1 carbon atom remaining in ethanoic acid to put into the formula. It should look like this:
C(1)H(2 x 1 + 1)COOH
CH3COOH
If you are happy with remembering to remove one atom from n when using this general formula, then since the sum in the formula is relatively straight forward, you should have no problems with it. On the other hand, the alternative general formula can be useful if you are prone to forgetting such details (its just a little more complex to remember):
Ethanoic acid n = 2
Cn-1H2(n-1)+1COOH
C2-1H2((2)-1)+1COOH
C1H2(1)+1COOH
CH3COOH
Aldehydes and Ketones are isomers of one another. Therefore, they will have the same formula in the form CnHmO. However, when writing their formulae, it is customary to differentiate them to indicate the presence of their functional group. For aldehydes, we include a -CHO functional group in the formula. This is not to be confused with a hydroxyl group (-OH, or -COH in this form).
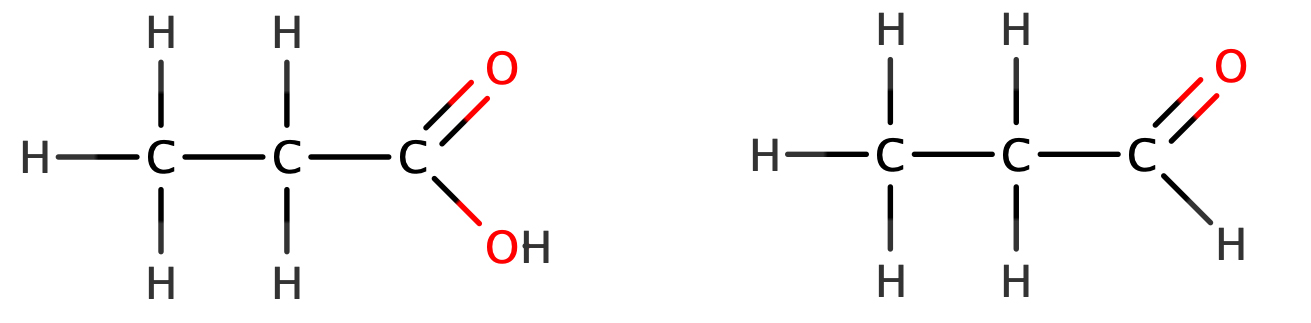
Look closely at the structures of propanoic acid and propanal. They both contain a carbonyl groups (-C=O). Think back to the reactions of aldehydes and ketones. Primary and secondary alcohols can be oxidised to aldehydes and ketones respectively; aldehydes can be oxidised further to carboxylic acids. The process of oxidation involves increasing the oxygen : hydrogen ratio. An aldehyde’s terminal carbon atom possesses bonds to one hydrogen atom and one double bond to an oxygen atom. Oxidising it removes the hydrogen and replaces it with a hydroxyl group – giving one single bond to an oxygen atom and one double bond to another oxygen atom.
This oxidation relationship between the structure of carboxylic acids and aldehydes is reflected in their chemical and general formulae. To differentiate between aldehydes and ketones, as well as indicate their functional groups being similar to carboxylic acids, aldehydes have the general formulae:
CnH2n+1CHO
Or
Cn-1H2(n-1)+1CHO
Once again, there are two possible general formulae to choose from. Remember – you don’t have to remember both, just pick the one you feel the most comfortable using.
So, with the example of propanal, where n = 3 :
C(2)H((2 x 2)+ 1)CHO
C2H5CHO
Remembering to save one carbon atom for the -CHO functional group
Or
C3-1H(2 x (3-1))+1CHO
C2H2 x (2)+1CHO
C2H5CHO
However, it should be noted that the general formula of aldehydes is often simplified to just CnH2nO, omitting any information about differentiating between aldehydes and ketones.
The ketones also have a functional group – the same as the aldehydes – the carbonyl group (-C=O). Since it is not located on a terminal carbon atom, the chemical formula of an aldehyde does not reveal its position, it simply contains an oxygen atom within it.
Propanal’s ketone isomer is called propanone:
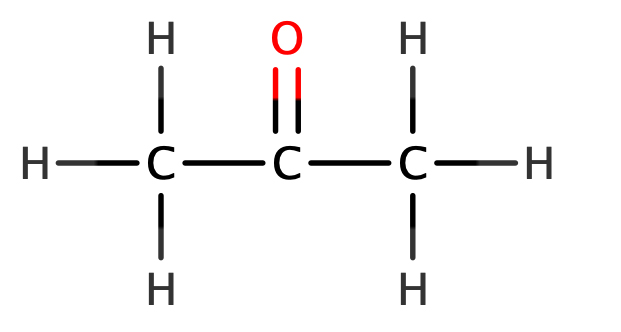
The general formula of ketones is:
CnH2nO
So for propanone, n = 3.
C(3)H2 x 3O
C3H6O
Note here that there is no need to subtract one carbon from the value of n since there is no functional group incorporated into the formula.
Application of General Formulae
General formulae can also serve you as a powerful tool for answering questions – particularly those asking you to write the chemical formula.
Let’s apply this to even larger molecules. Pentacontane is a large alkane with 50 carbon atoms, what would its formula be, and how many hydrogen atoms does it have?
We know from its name it is an alkane (the -ane suffix gives this away) and the Greek prefix pentacont- means 50. We simply have to put this number into the general formula for the alkanes:
CnH2n+2
C(50)H2(50)+2 = C50H102
Therefore, pentacontane it has 50 carbon atoms and 102 hydrogen atoms.
How about hectanoic acid? The hect- prefix means 100 carbon atoms, and it is clearly a carboxylic acid, so it must have a -COOH functional group, so it’s general formula is CnH2n+1COOH
Remembering that one of the carbon atoms forms part of the -COOH group, the formula must be:
C(99)H(2 x 99 + 1)COOH
C99H199COOH
It has 100 carbon atoms (the 99 being in the alkane chain and the remaining on in the COOH group) and 200 hydrogen atoms (once again, 199 being in the alkane chain and one left over on the -OH of the -COOH group).
It may seem like a very minor component of the organic chemistry units but being able to work out the general formula of a homologous series and being adept at using them to work out chemical formulae is a vital skill for all chemists. Learning where general formulae come from and how to use them properly will deepen pupil’s understanding of the structure of organic molecule’s.
If you would like further practice, or extra challenge with general formulae, try the questions in the worksheets linked below.
Rules for using the general formula:
- Identify the functional group (is it a carbon chain? Double bond? Carbonyl? COOH? Hydroxyl?)
- How many carbon atoms does it have? This gives you the value for n (however if it is an aldehyde or carboxylic acid, remember to either subtract 1 from this, or use the alternative general formulae)
- Enter the value for n (the number of carbon atoms) into the chosen general formula and calculate.
If you cannot remember the general formulae or need to work it out :
- Identify the functional group (is it a carbon chain? Double bond? Carbonyl? COOH? Hydroxyl?). Use this to assign the end of the formulae – OH, CHO, COOH, or just leave it as it is.
- How many carbon and hydrogen atoms does it have? Try to think how these two numbers relate to one another. Almost always you need to double the number of carbons to get the number of hydrogens – from here do you leave it as it is (2n), add one (2n+1), or add two (2n+2)? Consider as well whether one carbon atom is taken up in the functional group as with aldehydes and carboxylic acids. If so, reduce your value of nby one, or replace it in the general formula with n-1.